 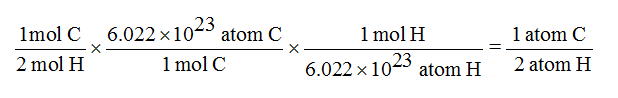
Please let us know how we can improve this web app. Related: Molecular weights of amino acids To calculate the mass of a single atom of carbon, we just need to divide the molar mass of 12.0 g (0,012 kg) by the number of particles per mole (Avogadros. 
The carbon dioxide gas is produced by the reaction of calcium carbonate with hydrochloric acid. In this experiment the molecular weight of carbon dioxide will be determined. Thus, Avogadro's number has been measured to be 6.022141×10. Thus, the molecular weight (or molar mass) of a gas can be determined by measuring the temperature, pressure, volume and the mass of a sample of the gas. Or, a little easier to understand, Avogadro's number is the ratio of 1 gram to 1 atomic mass unit but with the atomic mass unit expressed in grams. Weights of atoms and isotopes are from NIST article. Avogadro's number is the ratio of the mass of 12 grams of carbon-12 to the mass of 1 atom of carbon-12 measured in grams. If the molecular (or molar) mass of the substance is known. Weights of atoms and isotopes are from NIST article. The molar amounts of carbon and oxygen in a 100-g sample are calculated by dividing each element’s mass by its molar mass: 27.29 g C (mol C 12.01 g) 2.272 mol C 72.71 g O (mol O 16.00 g). 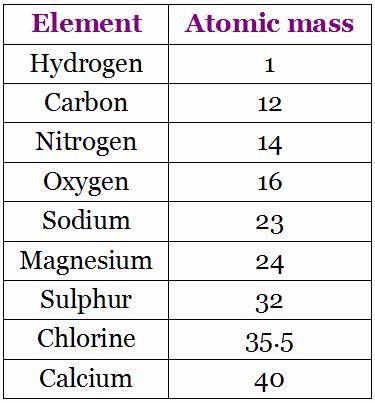
(1 u is equal to 1/12 the mass of one atom of carbon-12) Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol. A compound is 81.7 carbon and 18.3 hydrogen. Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u). Objective 3: Calculate the number of molecules, formula. The empirical formula of a compound is NH, and its molecular weight is 30.0 amu. Molar mass ( molar weight) is the mass of one mole of a substance and is expressed in g/mol. Objective 2: Use molar mass to convert between mass in grams and amount in moles of a chemical compound.The molar mass of formaldehyde is 30 g/mol, and at room temperature and. (1 u is equal to 1/12 the mass of one atom of carbon-12) Structural Formula of Formaldehyde Formaldehyde consists of two carbon where one. Molecular mass ( molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u).To calculate molecular weight of a chemical compound enter it's formula, specify its isotope mass number after each element in square brackets.Įxamples of molecular weight computations:ĭefinitions of molecular mass, molecular weight, molar mass and molar weight Molar mass calculator also displays common compound name, Hill formula, elemental composition, mass percent composition, atomic percent compositions and allows to convert from weight to number of moles and vice versa.Ĭomputing molecular weight (molecular mass) Functional groups: D, Ph, Me, Et, Bu, AcAc, For, Ts, Tos, Bz, TMS, tBu, Bzl, Bn, Dmg.Capitalize the first letter in chemical symbol and use lower case for the remaining letters: Ca, Fe, Mg, Mn, S, O, H, C, N, Na, K, Cl, Al. Computing molar mass (molar weight)To calculate molar mass of a chemical compound enter its formula and click 'Compute'. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
